 
The nitrogen in our DNA, the calcium in our teeth, the iron in our blood, the carbon in our apple pies were made in the interiors of collapsing stars. The data for the table came from OSU's Jennifer Johnson, who quotes Carl Sagan: Looking at the picture, you can see there are two electrons in shell one and four. The gold in your jewelry was likely made from neutron stars during collisions that may have been visible as short-duration gamma-ray bursts or gravitational wave events. Chalkboard with description of periodic table notation for carbon. Much of the iron in your body was made during supernovas of stars that occurred long ago and far away. The carbon in your body was made by nuclear fusion in the interior of stars, as was the oxygen. There are no other appreciable sources of hydrogen in the universe. The hydrogen in your body, present in every molecule of water, came from the Big Bang. was responsible for creating each element, according to our present understanding of the universe. It might actually play a critical role in the early chemical stages of the origins of life,” elaborated Olivier Berné of the French National Centre for Scientific Research in Toulouse, lead author of the study.From Wikipedia contributor Cmglee and Astronomy Picture of the Day, a color-coded periodic table that displays which cosmic events - the Big Bang, exploding stars, merging neutron stars, etc. For example, carbon has six protons and is atomic number 6. Symbol: a one or two letter symbol that represents the element. Atomic Number: the number of protons in the nucleus (which is the same as the number of electrons in the atom). “This clearly shows that ultraviolet radiation can completely change the chemistry of a protoplanetary disk. The number of protons in a nucleus determines the elements atomic number on the Periodic Table. Here's a close-up look at the carbon square from the Periodic Table. In particular, they could not detect any signs of water. Once formed, it then promotes additional chemical reactions to build more complex carbon molecules.īroadly, the team notes that the molecules they see in d203-506 are quite different from typical protoplanetary disks. However, the team predicts that UV radiation might actually provide the necessary source of energy for CH 3 + to form in the first place. Typically, UV radiation is expected to destroy complex organic molecules, in which case the discovery of CH 3 + might seem to be a surprise. 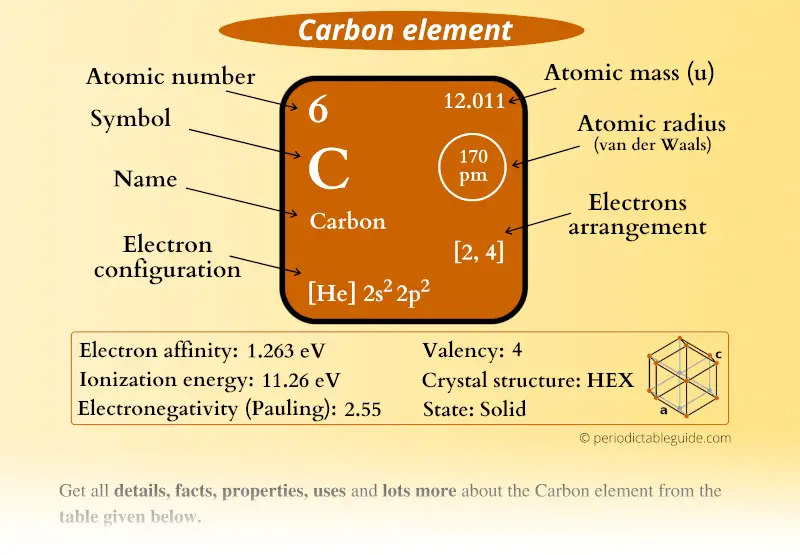
Scientists believe that most planet-forming disks go through a period of such intense UV radiation, since stars tend to form in groups that often include massive, UV-producing stars. 14 Carbon makes up about 0.025 percent of Earth's crust. It belongs to group 14 of the periodic table. It is nonmetallic and tetravalent its atom making four electrons available to form covalent chemical bonds. While the star in d203-506 is a small red dwarf, the system is bombarded by strong ultraviolet (UV) light from nearby hot, young, massive stars. Carbon (from Latin carbo 'coal') is a chemical element with the symbol C and atomic number 6. “This detection not only validates the incredible sensitivity of Webb but also confirms the postulated central importance of CH 3 + in interstellar chemistry,” said Marie-Aline Martin-Drumel of the University of Paris-Saclay in France, a member of the science team. In particular, Webb’s detection of a series of key emission lines from CH 3 + cemented the discovery. Carbon atoms can bond together in many different ways producing graphite. A carbon atom has six protons, six neutrons, and six electrons. Carbons atomic number is 6 and its symbol is C. Webb’s exquisite spatial and spectral resolution, as well as its sensitivity, all contributed to the team’s success. Based on the image, which statement is accurate A. The unique capabilities of Webb made it an ideal observatory to search for this crucial molecule. Carbon dioxide is a greenhouse gas, and it is dissolved in all natural waters. The study of interstellar organic (carbon-containing) chemistry, which Webb is opening in new ways, is an area of keen fascination to many astronomers. Keeling Curve Carbon is present as carbon dioxide in Earth’s atmosphere at a concentration of about 0.04 percent by volume, an amount that is increasing. Methyl cation was detected in a young star system, with a protoplanetary disk, known as d203-506, which is located about 1,350 light-years away in the Orion Nebula.Ĭarbon compounds form the foundations of all known life, and as such are particularly interesting to scientists working to understand both how life developed on Earth, and how it could potentially develop elsewhere in our universe. Known as methyl cation (pronounced cat-eye-on) (CH 3 +), the molecule is important because it aids the formation of more complex carbon-based molecules. A team of international scientists has used NASA’s James Webb Space Telescope to detect a new carbon compound in space for the first time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
